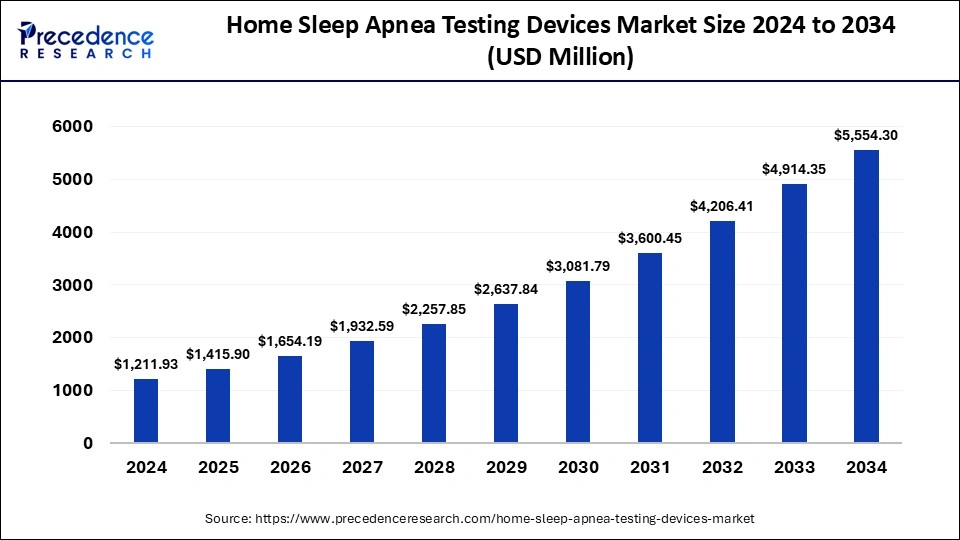
The global home sleep apnea testing devices market was valued at USD 1,037.34 million in 2023 and is expected to reach around USD 4,914.35 million by 2033. The market is expanding at a solid CAGR of 16.83% over the forecast period 2024 to 2033.
Key Points
- The North America home sleep apnea testing devices market size accounted for USD 352.70 million in 2023 and is expected to attain around USD 1,695.45 million by 2033, poised to grow at a CAGR of 17% between 2024 and 2033.
- North America has held a major revenue share of 34% in 2023.
- Asia Pacific is projected to experience the fastest growth in the market during the forecast period.
- By type, the type 3 segment dominated the market with the largest revenue share of 72% in 2023.
- By type, the type 4 segment is expected to witness the fastest growth in the market during the forecast period.
The home sleep apnea testing devices market has witnessed significant growth over the past decade, driven by increasing awareness of sleep apnea and its associated health risks. Sleep apnea is a common disorder characterized by interruptions in breathing during sleep, leading to poor sleep quality and various health issues such as cardiovascular diseases, diabetes, and hypertension. Traditionally, diagnosis and monitoring of sleep apnea were conducted in sleep laboratories or clinics. However, the advent of home sleep apnea testing (HSAT) devices has revolutionized the approach to diagnosing this condition by offering a more convenient, cost-effective, and accessible alternative.
Home sleep apnea testing devices are portable, easy-to-use equipment designed to record various physiological parameters such as airflow, respiratory effort, blood oxygen levels, and heart rate. These devices allow patients to undergo sleep studies in the comfort of their own homes, enhancing patient compliance and facilitating early diagnosis and treatment. The market for HSAT devices encompasses a wide range of products, including wearable devices, finger oximeters, and integrated systems with multiple sensors.
Get a Sample: https://www.precedenceresearch.com/sample/4426
Growth Factors
Several key factors contribute to the growth of the home sleep apnea testing devices market:
- Rising Prevalence of Sleep Disorders: The global prevalence of sleep apnea and other sleep-related disorders has been steadily increasing. Sedentary lifestyles, obesity, and aging populations are significant contributors to the rising incidence of sleep apnea, driving the demand for diagnostic solutions like HSAT devices.
- Technological Advancements: Continuous advancements in technology have led to the development of more accurate, user-friendly, and affordable home sleep apnea testing devices. Innovations such as wireless connectivity, cloud-based data storage, and AI-driven analysis enhance the functionality and reliability of these devices.
- Cost-Effectiveness and Convenience: Home sleep apnea testing offers a cost-effective alternative to traditional polysomnography conducted in sleep labs. It eliminates the need for overnight stays in medical facilities, reducing healthcare costs and providing greater convenience for patients.
- Increasing Awareness and Education: Efforts by healthcare organizations and advocacy groups to raise awareness about the dangers of untreated sleep apnea have led to increased demand for diagnostic testing. Public health campaigns and education initiatives emphasize the importance of early detection and treatment.
- Favorable Reimbursement Policies: In many regions, healthcare insurance providers have started covering home sleep apnea testing, making it more accessible to a broader population. Favorable reimbursement policies encourage patients and healthcare providers to adopt HSAT devices.
Region Insights
The home sleep apnea testing devices market exhibits regional variations in terms of adoption rates, market dynamics, and growth potential. Key regional insights include:
- North America: North America, particularly the United States, dominates the HSAT devices market. High awareness levels, advanced healthcare infrastructure, and a large patient pool with sleep apnea contribute to the region’s market leadership. Additionally, supportive reimbursement policies and the presence of leading market players drive growth in this region.
- Europe: Europe holds a significant share of the global HSAT devices market, with countries like Germany, the United Kingdom, and France being major contributors. The growing geriatric population, increasing obesity rates, and well-established healthcare systems support market expansion. Government initiatives and public health campaigns also play a crucial role in raising awareness about sleep disorders.
- Asia-Pacific: The Asia-Pacific region is experiencing rapid growth in the home sleep apnea testing devices market. Factors such as improving healthcare infrastructure, rising disposable incomes, and increasing awareness about sleep apnea are driving demand. Countries like China, Japan, and India offer substantial growth opportunities due to their large populations and growing middle class.
- Latin America: Latin America is emerging as a promising market for HSAT devices, with countries like Brazil and Mexico leading the way. Growing urbanization, increasing prevalence of sleep disorders, and expanding healthcare access contribute to market growth. However, economic disparities and limited healthcare resources in some areas pose challenges.
- Middle East and Africa: The Middle East and Africa region shows potential for market expansion, particularly in Gulf Cooperation Council (GCC) countries. Rising healthcare investments, increasing awareness, and a growing focus on preventive healthcare drive demand for HSAT devices. However, limited healthcare infrastructure and economic constraints in certain regions may hinder growth.
Home Sleep Apnea Testing Devices Market Scope
| Report Coverage | Details |
| Home Sleep Apnea Testing Devices Market Size in 2023 | USD 1,037.34 Million |
| Home Sleep Apnea Testing Devices Market Size in 2024 | USD 1,211.93 Million |
| Home Sleep Apnea Testing Devices Market Size by 2033 | USD 4,914.35 Million |
| Home Sleep Apnea Testing Devices Market Growth Rate | CAGR of 16.83% from 2024 to 2033 |
| Largest Market | North America |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Home Sleep Apnea Testing Devices Market Dynamics
Drivers
The home sleep apnea testing devices market is driven by several factors that collectively enhance its growth trajectory:
- Growing Burden of Sleep Apnea: The increasing prevalence of sleep apnea, particularly obstructive sleep apnea (OSA), is a significant driver. As more individuals seek diagnosis and treatment for this condition, the demand for HSAT devices rises correspondingly.
- Shift Towards Home-Based Healthcare: The trend towards home-based healthcare solutions, accelerated by the COVID-19 pandemic, has boosted the adoption of HSAT devices. Patients prefer home testing due to its convenience, comfort, and reduced risk of exposure to infectious diseases.
- Healthcare Cost Containment: Healthcare systems worldwide are under pressure to contain costs while providing effective care. Home sleep apnea testing offers a cost-effective alternative to traditional in-lab polysomnography, reducing overall healthcare expenses and making it an attractive option for both patients and providers.
- Technological Innovations: Continuous advancements in sensor technology, data analytics, and wireless communication have enhanced the accuracy and ease of use of HSAT devices. Features such as remote monitoring and cloud-based data storage improve patient engagement and streamline diagnostic processes.
- Supportive Government Policies: Government initiatives and policies that promote early diagnosis and management of sleep disorders contribute to market growth. Public health campaigns and educational programs increase awareness and encourage individuals to seek testing and treatment.
Opportunities
The home sleep apnea testing devices market presents several opportunities for growth and innovation:
- Expansion into Emerging Markets: Emerging markets in Asia, Latin America, and Africa offer significant growth potential due to increasing healthcare investments, rising disposable incomes, and growing awareness about sleep disorders. Market players can tap into these regions by introducing affordable and accessible HSAT devices.
- Integration with Telemedicine: The integration of HSAT devices with telemedicine platforms can enhance patient care by enabling remote consultations, monitoring, and data sharing with healthcare providers. This approach improves patient outcomes and increases the adoption of home-based testing solutions.
- Development of Wearable Devices: Wearable HSAT devices that are comfortable and easy to use can enhance patient compliance and broaden the market. Innovations in wearable technology, such as smartwatches and fitness trackers with integrated sleep monitoring features, can drive market growth.
- Personalized Healthcare Solutions: Advances in data analytics and artificial intelligence can enable personalized healthcare solutions for sleep apnea patients. Tailoring treatment plans based on individual patient data can improve the effectiveness of interventions and enhance patient satisfaction.
- Collaborations and Partnerships: Collaborations between device manufacturers, healthcare providers, and research institutions can drive innovation and market expansion. Joint ventures and partnerships can facilitate the development of advanced HSAT devices and improve market penetration.
Challenges
Despite the promising growth prospects, the home sleep apnea testing devices market faces several challenges:
- Regulatory Hurdles: Regulatory approval processes for medical devices can be complex and time-consuming. Compliance with stringent regulations and standards is essential to ensure the safety and efficacy of HSAT devices, posing challenges for manufacturers.
- Limited Awareness and Education: While awareness about sleep apnea is increasing, there is still a lack of widespread understanding among the general population. Efforts to educate patients, primary care physicians, and healthcare providers about the benefits of home sleep apnea testing are crucial.
- Data Privacy and Security Concerns: The use of HSAT devices involves the collection and transmission of sensitive patient data. Ensuring data privacy and security is paramount to gain patient trust and comply with regulatory requirements. Data breaches and cybersecurity threats pose significant risks.
- Accuracy and Reliability Issues: Although technological advancements have improved the accuracy of HSAT devices, concerns about their reliability compared to traditional polysomnography remain. Ensuring that home testing provides accurate and consistent results is essential for widespread adoption.
- Reimbursement Challenges: While reimbursement policies for HSAT devices have improved, there are still challenges related to coverage and reimbursement rates. Variability in insurance policies and coverage criteria can impact patient access to home testing solutions.
Read Also: Endoscope Reprocessing Market Size to Reach USD 3.79 Bn By 2033
Home Sleep Apnea Testing Devices Market Companies
- Fisher & Paykel Healthcare
- ImThera Medical, Inc.
- Natus Medical, Inc.
- Somnetics International, Inc.
- Braebon Medical Corporation
- BMC Medical Co., Ltd.
- Teleflex, Inc.
- Weinmann Medical Devices GmbH & Co., KG.
- Philips Respironics
- CareFusion Corp.
- Compumedics Limited
- and Nihon Kohden.
Home Sleep Apnea Testing Devices Market Recent Developments
- In February 2023, Philips Respironics introduced the release of its new domestic sleep screening tool, the Philips DreamMapper Pro. The DreamMapper Pro is a software platform that may be used to tune and control sleep apnea remedies.
- In March 2023, SomnoMed introduced the release of its new domestic sleep screening tool, the SomnoMed AXG300. The AXG300 is a wi-fi device that can be used to diagnose sleep apnea and loud night breathing.
- In April 2023, Welch Allyn introduced the release of its new domestic sleep screening tool, the Welch Allyn SleepCheck Pro. The SleepCheck Pro is a fingertip pulse oximeter that may be used to screen blood oxygen degrees all through sleep.
- In November 2022, ResMed and Alphabet’s life science offshoot Verily announced the formation of Primasun, an end-to-end solution to help employers and healthcare providers identify populations at risk for complex sleep disorders.
- In October 2022, Airway Management, manufacturer of the most globally researched custom oral appliances, announced the launch of flexTAP, a premium lab-made oral appliance designed to treat patients with snoring and mild to moderate obstructive sleep apnea.
Segments Covered in the Report
By Test Type
- Type 2
- Type 3
- Type 4
By Geography
- North America
- Asia Pacific
- Europe
- Latin America
- Middle East & Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/