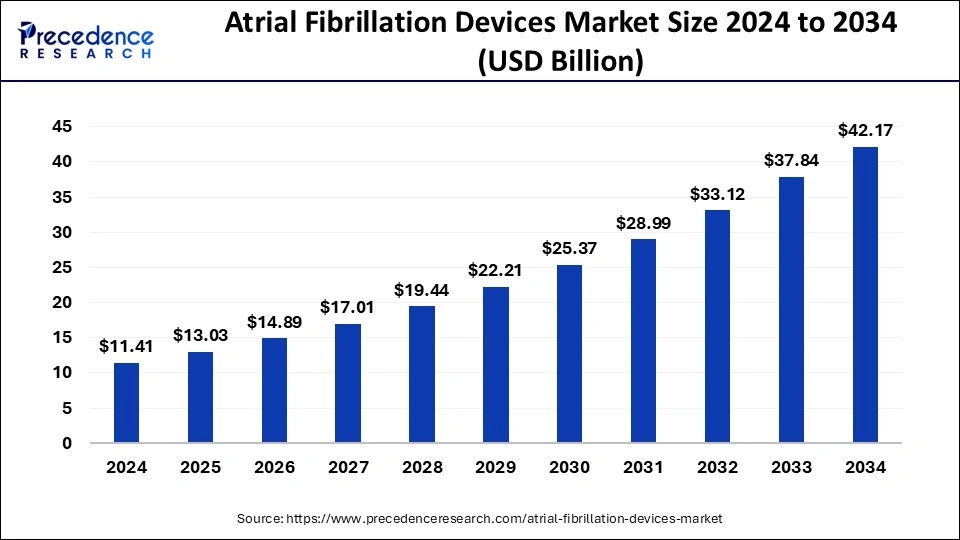
The global atrial fibrillation devices market size is estimated to attain around USD 37.84 billion by 2033, growing at a CAGR of 14.25% from 2024 to 2033.
Key Points
- North America dominated the market share of 40% in 2023.
- By product, the EP ablation catheters segment dominated the atrial fibrillation devices market in 2023.
- By end-use, the hospitals segment dominated the market in 2023.
The Atrial Fibrillation Devices Market refers to the sector encompassing medical devices specifically designed for the treatment and management of atrial fibrillation (AF), a common heart rhythm disorder characterized by irregular and often rapid heartbeats. These devices are crucial in providing therapeutic interventions to patients suffering from AF, aiming to restore normal heart rhythm, alleviate symptoms, and reduce the risk of complications associated with the condition. The market for AF devices has witnessed significant growth in recent years due to the rising prevalence of AF globally, advancements in technology, and increasing demand for minimally invasive treatment options.
Get a Sample: https://www.precedenceresearch.com/sample/3950
Growth Factors:
Several key factors contribute to the growth of the Atrial Fibrillation Devices Market. Firstly, the growing prevalence of atrial fibrillation worldwide has led to an increased demand for effective treatment options. With an aging population and rising incidence of risk factors such as hypertension, obesity, and diabetes, the number of AF cases is expected to rise, driving market growth. Additionally, technological advancements in AF devices, such as the development of catheter-based ablation techniques and the introduction of novel implantable devices, have enhanced treatment efficacy and safety, further fueling market expansion. Moreover, the increasing adoption of minimally invasive procedures, shorter hospital stays, and quicker recovery times have boosted patient acceptance and contributed to market growth.
Region Insights:
The Atrial Fibrillation Devices Market exhibits regional variations in terms of market size, growth rates, and key market players. North America and Europe dominate the market, owing to factors such as well-established healthcare infrastructure, high adoption rates of advanced medical technologies, and favorable reimbursement policies. These regions also witness significant investments in research and development activities, driving innovation in AF device technologies. In contrast, the Asia-Pacific region is experiencing rapid market growth attributed to increasing healthcare expenditure, growing awareness about AF diagnosis and treatment, and rising prevalence of risk factors predisposing individuals to AF, such as hypertension and diabetes. Emerging economies in Asia-Pacific, including China and India, present lucrative opportunities for market players due to large patient populations and improving access to healthcare services.
Atrial Fibrillation Devices Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 14.25% |
| Global Market Size in 2023 | USD 9.99 Billion |
| Global Market Size by 2033 | USD 37.84 Billion |
| U.S. Market Size in 2023 | USD 3 Billion |
| U.S. Market Size by 2033 | USD 11.35 Billion |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Product and By End-use |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
Atrial Fibrillation Devices Market Dynamics
Drivers:
Several drivers contribute to the growth of the Atrial Fibrillation Devices Market. Firstly, the aging population worldwide, coupled with the increasing prevalence of lifestyle-related risk factors, such as obesity and hypertension, has led to a higher incidence of atrial fibrillation, driving demand for AF devices. Additionally, the shift towards minimally invasive procedures, such as catheter ablation and left atrial appendage closure, has resulted in shorter hospital stays, reduced complications, and improved patient outcomes, thereby boosting market growth. Furthermore, the growing focus on preventive healthcare and the introduction of novel technologies, such as robotic-assisted ablation systems and advanced mapping technologies, have expanded the treatment options available for AF patients, stimulating market expansion.
Opportunities:
The Atrial Fibrillation Devices Market presents numerous opportunities for growth and innovation. One such opportunity lies in the development of next-generation AF devices with enhanced safety, efficacy, and procedural outcomes. Market players can capitalize on advancements in materials science, robotics, and artificial intelligence to develop novel devices that offer precise lesion formation, real-time navigation, and personalized treatment approaches. Moreover, expanding market penetration in emerging economies, investing in physician training programs, and strengthening distribution networks can unlock new growth avenues for AF device manufacturers. Additionally, strategic collaborations and partnerships with healthcare providers, research institutions, and regulatory bodies can facilitate the development and commercialization of innovative AF solutions, further driving market expansion.
Challenges:
Despite the promising growth prospects, the Atrial Fibrillation Devices Market faces several challenges that warrant attention. One significant challenge is the stringent regulatory requirements governing the approval and commercialization of AF devices, which often entail lengthy and costly clinical trials. Moreover, reimbursement challenges and pricing pressures in healthcare systems worldwide pose barriers to market entry for smaller companies and hinder widespread adoption of AF devices. Additionally, the complex nature of atrial fibrillation, variability in patient anatomy, and the risk of procedural complications necessitate ongoing advancements in device technologies and operator skills to improve treatment outcomes and patient safety. Furthermore, market competition and consolidation among key players intensify competitive pressures, requiring companies to differentiate their offerings and innovate continuously to maintain market share and sustain growth.
Read Also: Cardiac Arrhythmia Monitoring Devices Market Size, Report 2033
Recent Developments
- In November 2023, Medtronic introduced a heart implant to reduce the lifetime risk of stroke in patients with atrial fibrillation and improve the quality of life for patients undergoing open cardiac surgery.
- In January 2022, AliveCor, Inc. and Voluntis, a leading Aptar Pharma firm in digital therapies, have partnered to provide advanced management of atrial fibrillation for cancer patients.
Atrial Fibrillation Devices Market Companies
- Abbott Laboratories
- Johnson & Johnson
- Atricure Inc
- Microport Scientific Corporation
- Boston Scientific Corporation
- St. Jude Medical, Inc
- Medtronic Plc
- Koninklijke Philips N.V.
- Siemens AG
Segments Covered in the Report
By Product
- EP Ablation Catheters
- EP Diagnostic Catheters
- Mapping and Recording Systems
- Cardiac Monitors or Implantable Loop Recorder
- Access Devices
- Intracardiac Echocardiography (ICE)
- Left Atrial Appendage (LAA) Closure Devices
By End-use
- Hospitals
- Cardiac Centers
- Ambulatory Surgical Centers
- Others
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/