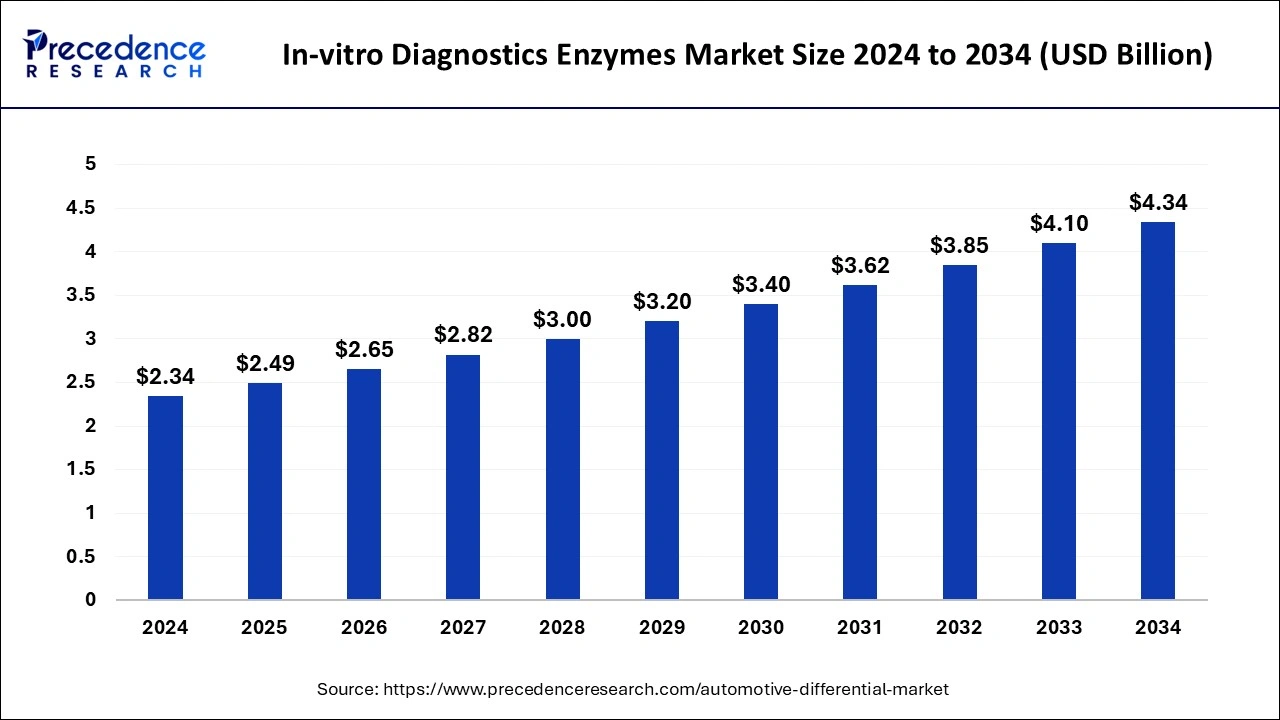
The global in-vitro diagnostics enzymes market size is estimated to grow around USD 4.10 billion by 2033, growing at a CAGR of 6.42% from 2024 to 2033.
Key Points
- The North America in-vitro diagnostics enzymes market size was estimated at USD 920 million in 2023 and is projected to surpass around USD 1,720 million by 2033.
- North America dominated the in-vitro diagnostics enzymes market in 2023 with a revenue share of 42%.
- Asia Pacific is expected to grow at the fastest rate during the forecast period.
- By enzyme type, the polymerase and transcriptase segment has captured a market share of 37% in 2023.
- By end-user, the hospitals and diagnostic segment has held a market share of over 41.8% in 2023.
- By technology, the histology assays segment has accounted market share of 44% in 2023.
- By disease, the infectious disease segment dominated the market in 2023.
The in-vitro diagnostics (IVD) enzymes market involves the production and use of enzymes for diagnostic tests conducted in a controlled environment, such as a laboratory. These tests help in detecting and monitoring diseases, medical conditions, and overall health. The market is seeing significant growth due to the rising demand for diagnostic tests in various healthcare settings.
Get a Sample: https://www.precedenceresearch.com/sample/4095
Growth Factors
The growth of the in-vitro diagnostics enzymes market is driven by an increasing prevalence of chronic diseases such as diabetes and cancer, which require regular monitoring and testing. Additionally, advancements in enzyme technology, coupled with the growing use of point-of-care testing, contribute to market expansion. Increased awareness of preventive healthcare and early diagnosis further fuels the demand for IVD enzymes.
Regional Insights:
North America leads the IVD enzymes market due to a well-established healthcare infrastructure, high healthcare expenditure, and rapid adoption of advanced diagnostic technologies. Europe follows closely, with a growing focus on preventive healthcare and increasing investments in research and development. The Asia-Pacific region is experiencing rapid growth, driven by an expanding population, rising disposable incomes, and a surge in healthcare investments.
In-vitro Diagnostics Enzymes Market Scope
| Report Coverage | Details |
| Growth Rate from 2024 to 2033 | CAGR of 6.42% |
| Global Market Size in 2023 | USD 2.20 Billion |
| Global Market Size in 2024 | USD 2.34 Billion |
| Global Market Size by 2033 | USD 4.10 Billion |
| Largest Market | North America |
| Base Year | 2023 |
| Forecast Period | 2024 to 2033 |
| Segments Covered | By Enzyme Type, By Disease Type, By Technology Type, and By End-use |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
In-vitro Diagnostics Enzymes Market Dynamics
Drivers:
Key drivers for the IVD enzymes market include technological advancements in diagnostic tools, increasing demand for personalized medicine, and a surge in the number of point-of-care tests. Additionally, regulatory support and funding for research and development activities play a significant role in driving market growth.
Opportunities:
Opportunities in the IVD enzymes market arise from the growing trend towards home healthcare and telemedicine, which expands the use of point-of-care testing. Moreover, the development of novel diagnostic tests using enzymes offers potential for new applications and market expansion. Collaborations between research institutions and industry players also present growth prospects.
Challenges:
The IVD enzymes market faces challenges such as stringent regulatory requirements and high development costs, which can impede market entry for new players. Additionally, the availability of alternative diagnostic technologies and the risk of commoditization of enzymes may pose challenges to market growth. Addressing these challenges requires ongoing innovation and adaptation to changing market dynamics.
Read Also: Electronic Adhesives Market Size to Grow USD 12.40 Bn by 2033
Recent Developments
- In June 2023, GenWorks Health, a Bengaluru-based startup, launched an IVD test for malaria and dengue. This test includes rapid card test kits.
- In September 2023, NeoDX Biotech lab launched an IVD kit for detecting autoimmune disorders. It’s a real-time PCR- a technology-based kit to improve healthcare service and enhance testing abilities.
In-vitro Diagnostics Enzymes Market Companies
- Biocatalysts Ltd.
- Amicogen
- Dyadic International
- BBI Solutions
- Affymetrix
- American Laboratories
- Merck KGaA
- Codexis, Inc.
- F. Hoffmann-La Roche Ltd.
- Amano Enzyme Inc.
- Advanced Enzymes Technologies Ltd.
Segments Covered in the Report
By Enzyme Type
- Proteases
- Polymerase & Transcriptase
- Ribonuclease
- Others
BY Disease Type
- Infectious Disease
- COVID-19 Testing
- Hepatitis
- HIV
- Others
- Diabetes
- Oncology
- Cardiology
- Nephrology
- Autoimmune diseases
- Others
By Technology Type
- Histology Assays
- Molecular Diagnostics
- PCR Assays
- NGS Assays
- Others
- Clinical Chemistry
By End-use
- Pharma & Biotech
- Hospital & Diagnostic Labs
- Contract Research Organizations (CROs)
- Academic Labs
By Geography
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Contact Us:
Mr. Alex
Sales Manager
Call: +1 9197 992 333
Email: sales@precedenceresearch.com
Web: https://www.precedenceresearch.com
Blog: https://www.expresswebwire.com/
Blog: https://www.uswebwire.com/
Blog: https://www.dailytechbulletin.com/
Blog: https://www.autoindustrybulletin.com/