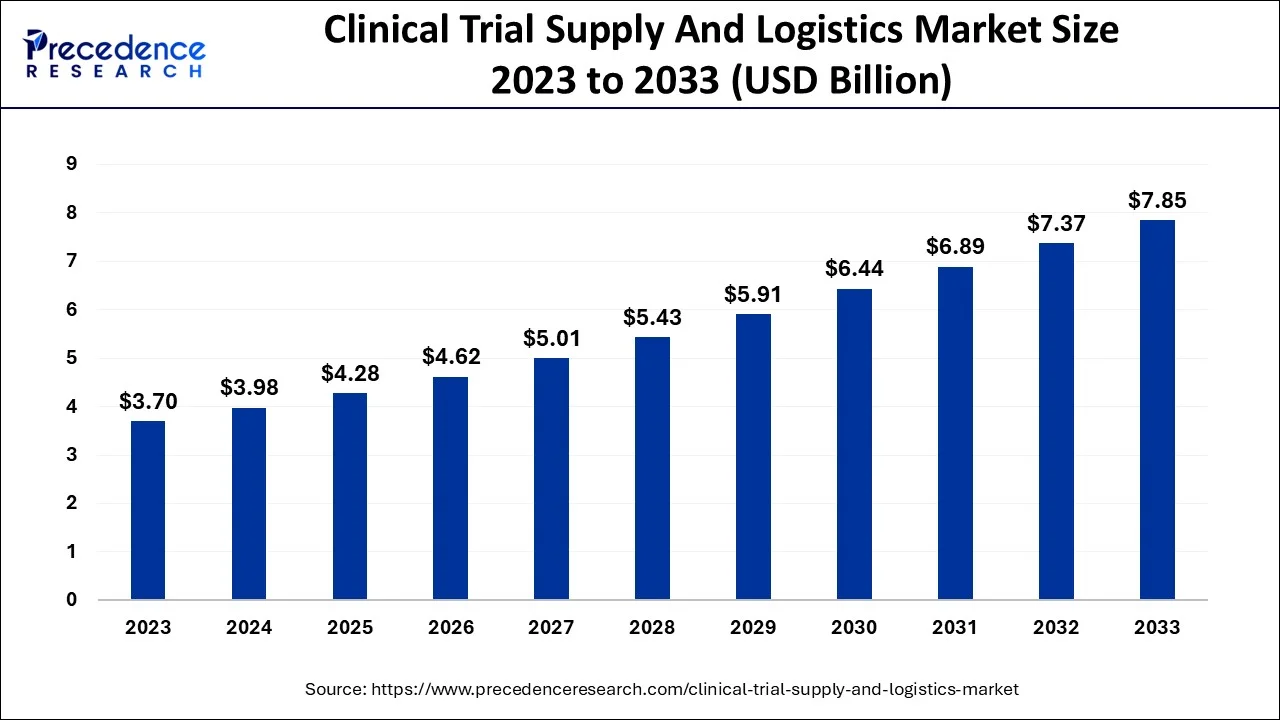
The global clinical trial supply and logistics market was valued at USD 3.98 billion in 2024 and is projected to grow to approximately USD 8.45 billion by 2034, expanding at a CAGR of 7.84% from 2025 to 2034. The market is driven by increasing complexity in clinical trials, globalization of research, and a growing pipeline of biologics, orphan drugs, and advanced therapies requiring specialized supply chain solutions.
Read Also: Ophthalmic Packaging Market
Key Market Opportunities
1. Expansion in Emerging Markets
-
Asia Pacific, Latin America, and Eastern Europe are seeing a surge in clinical trial activity due to large patient populations, cost advantages, and diverse disease demographics.
-
Opportunities exist for logistics providers to establish regional hubs and streamlined distribution networks to serve growing trial demand in these regions.
2. Rare Diseases and Orphan Indications
-
Increasing investment in precision medicine, orphan drugs, and rare disease research is creating demand for specialized supply chains.
-
Trials for these therapies often involve small, geographically dispersed patient populations, requiring flexible and adaptive logistics solutions.
3. Growth of Biologics and Advanced Therapies
-
The rise of cell and gene therapies, CAR-T therapies, and monoclonal antibodies necessitates ultra-low temperature storage, advanced cold chain logistics, and precise handling.
-
Providers offering end-to-end biologic logistics have a significant growth opportunity due to these specialized requirements.
4. Decentralized Clinical Trials (DCTs)
-
Collaborations such as Thermo Fisher Scientific and Matrix Clinical Trials indicate a shift toward patient-centric decentralized trials.
-
Opportunities exist in home delivery of investigational products, remote monitoring, and digital supply chain management for DCTs.
5. Technological Integration
-
Adoption of IoT-enabled tracking, real-time monitoring, blockchain for traceability, and AI-driven logistics optimization provides efficiency and compliance advantages.
-
Companies investing in digital and automated solutions can reduce delays, ensure regulatory compliance, and improve supply chain visibility.
What’s Driving Market Growth?
Increasing Complexity of Clinical Trials
The surge in biologics, CAR-T therapies, and precision medicines necessitates temperature-controlled storage, specialized packaging, and real-time monitoring. Multi-geography trials targeting rare diseases require adaptive and resilient supply chains, driving demand for advanced logistics services.
Globalization of Clinical Research
Emerging markets in Asia Pacific, Latin America, and Eastern Europe are experiencing rapid growth in clinical trial activity. Companies are leveraging regional hubs and digital coordination platforms to manage shipments across multiple countries, boosting the need for robust supply and logistics solutions.
Challenges in the Market
-
High Cost of Cold Chain Logistics: Ultra-low and cryogenic storage for biologics increases operational expenditure.
-
Regulatory Complexity: Diverse regulations across countries complicate labeling, import/export clearance, and compliance, posing risks to trial timelines.
Opportunities: Collaborations and Decentralized Trials
Increasing partnerships among market leaders are shaping growth. For example, Thermo Fisher Scientific Inc. and Matrix Clinical Trials are collaborating to develop innovative decentralized clinical trial (DCT) solutions in the U.S., enhancing patient recruitment and retention.
In-Depth Market Opportunities Analysis
1. Geographic Expansion & Emerging Market Potential
-
Asia Pacific, Latin America, and Eastern Europe are experiencing rapid growth in clinical trial activity.
-
Drivers:
-
Large patient pools: Easier recruitment and faster enrollment, especially for rare diseases.
-
Cost advantages: Lower operational and trial execution costs than in North America and Europe.
-
Government initiatives: Some countries offer regulatory incentives for clinical trials, such as faster approval timelines and tax benefits.
-
-
Opportunities:
-
Establish regional distribution hubs to streamline logistics for multi-site trials.
-
Offer customized supply chain solutions that account for local regulations, temperature control needs, and transportation infrastructure.
-
2. Rare Diseases & Orphan Drug Trials
-
Rare disease treatments are growing due to advances in genomics and personalized medicine.
-
Challenges in this space:
-
Small, dispersed patient populations.
-
High sensitivity of biologic materials.
-
-
Opportunities:
-
Specialized cold chain and packaging solutions.
-
Agile supply chains capable of quick scaling and custom batch shipments.
-
Partnerships with research hospitals and CROs to ensure timely delivery to scattered trial sites.
-
3. Biologics, Advanced Therapies & Personalized Medicine
-
Biologics, CAR-T therapies, and gene therapies dominate the innovation pipeline.
-
Requirements:
-
Ultra-low temperature logistics (cryogenic or -80°C storage).
-
Secure tracking and monitoring for product integrity.
-
Compliance with stringent regulations across multiple jurisdictions.
-
-
Opportunities:
-
Providers with advanced cold chain solutions can differentiate themselves.
-
High barriers to entry create premium service offerings for sensitive products.
-
Integration of real-time monitoring and IoT-enabled devices to provide end-to-end transparency.
-
4. Decentralized Clinical Trials (DCTs)
-
A growing trend toward patient-centric trials where participants can receive medications at home.
-
Opportunities:
-
Develop direct-to-patient distribution networks with robust tracking systems.
-
Innovative packaging and delivery solutions ensuring dosage accuracy, temperature stability, and tamper-proofing.
-
Digital integration with apps and monitoring platforms for compliance and real-time feedback.
-
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com