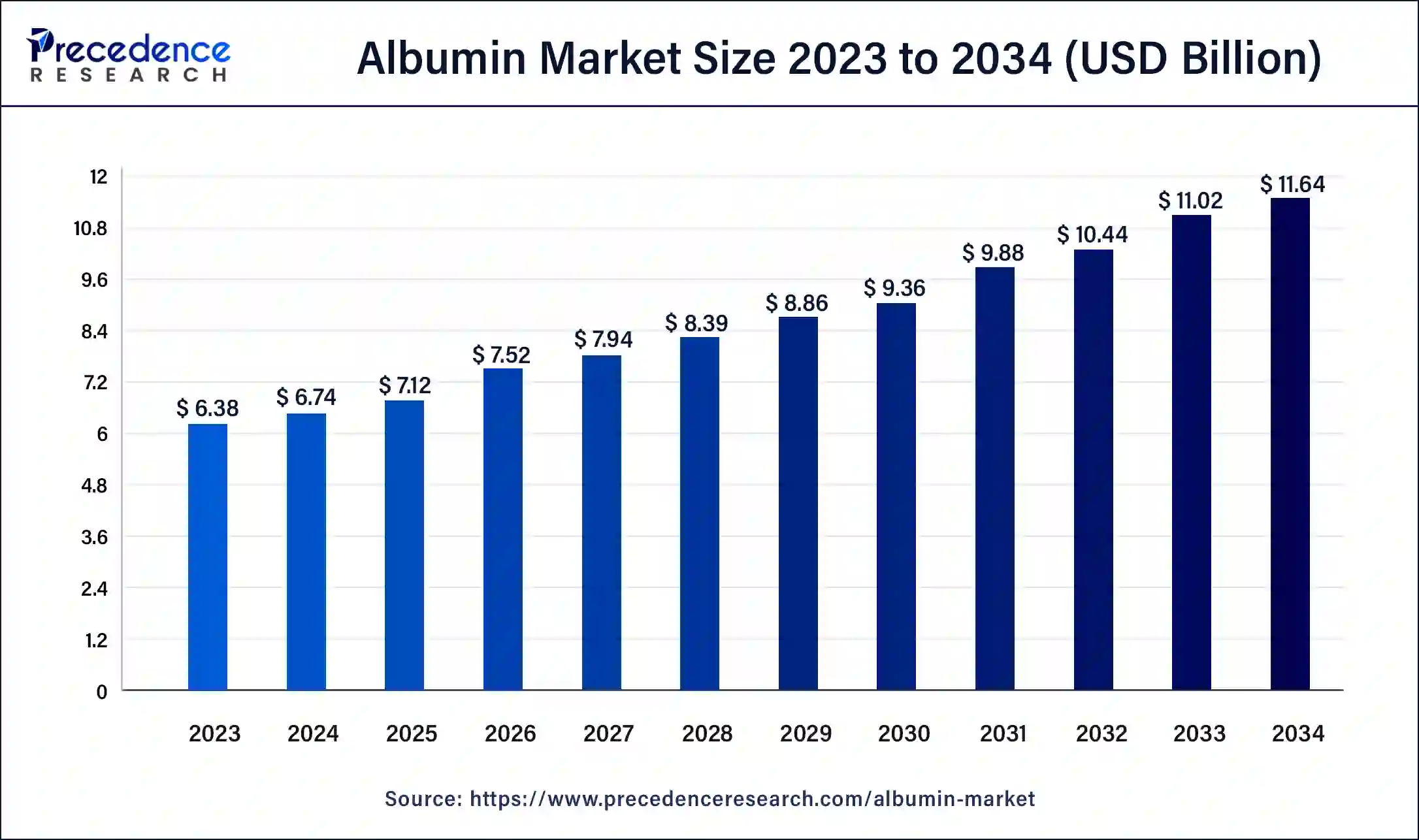
The global albumin market is gaining momentum, fueled by advancements in biotechnology, growing demand in emerging economies, and an increase in chronic disease cases. Valued at USD 6.74 billion in 2024, the market is expected to expand at a CAGR of 5.62% and reach USD 11.64 billion by 2034. With increasing applications in therapeutics, drug formulation, and diagnostics, albumin continues to be a critical component across the global healthcare sector.
Key Takeaways
-
Market Size: Expected to grow from USD 6.74 billion in 2024 to USD 11.64 billion by 2034.
-
Growth Rate: CAGR of 5.62% from 2025 to 2034.
-
Regional Highlights: North America held the largest share in 2024; Asia Pacific to witness the fastest growth.
-
Leading Segments:
-
Product: Human serum albumin dominated in 2024.
-
Application: Therapeutics segment held the largest share.
-
End-use: Hospitals and clinics dominated in 2024.
-
Read Also : Commercial Robots Market
Market Overview
Albumin, a protein produced by the liver and found abundantly in plasma, plays a vital role in maintaining oncotic pressure and facilitating various physiological processes. It is used in numerous clinical situations including volume resuscitation, liver diseases, and during surgeries. Its rising significance in therapeutic formulations and biotechnological innovations is bolstering market growth.
Market Drivers
Rising Cases of Hypoalbuminemia and Liver Diseases
The growing prevalence of hypoalbuminemia—a condition often associated with inflammation and chronic illness—is significantly driving demand for albumin therapies. According to the American Liver Foundation, over 100 million Americans are affected by some form of liver disease. This is creating a consistent need for albumin, particularly in the treatment of advanced liver conditions.
Expansion of Plasma Fractionation Facilities
Global investments in plasma fractionation infrastructure are propelling market growth. Recent developments include:
-
CSL’s plasma fractionation facilities launched in Germany (2023) and Australia (2022) to produce albumin-based medicines.
-
Enhanced automation and optimized processes are helping improve the safety, efficiency, and scalability of albumin extraction.
Market Challenges
High Production Costs and Regulatory Hurdles
Producing albumin, especially from human plasma, is costly and involves strict regulatory oversight. Compliance with FDA standards and industry regulations such as those from the Plasma Protein Therapeutic Association (PPTA) can be resource-intensive, potentially hindering smaller manufacturers.
Emerging Opportunities
Breakthroughs in Recombinant DNA Technology
Recombinant human albumin (rHA) is transforming the market. It offers a scalable, consistent, and safe alternative to plasma-derived albumin:
-
Shilpa Medicare filed its first Drug Master File for rHA with the U.S. FDA in June 2024.
-
Wuhan Healthgen Biotechnology Corp completed Phase III trials for plant-derived rHA in February 2024.
These innovations are set to reduce dependency on human donors and streamline albumin production globally.
Product Insights
-
Human Serum Albumin (HSA) remains the dominant product due to its clinical relevance and established efficacy in liver treatments and plasma expansion.
-
Bovine Serum Albumin (BSA) is gaining traction in lab research and diagnostic applications, with increased demand for animal-free and structurally identical alternatives.
Application Insights
-
Therapeutics lead the market, driven by albumin’s role in fluid replacement therapy, liver disease management, and critical care.
-
Drug Formulation is a fast-growing segment, with albumin enhancing drug solubility, stability, and delivery mechanisms. Notably, Panacea Biotec launched albumin-bound injectable particles in Canada in 2023 for cancer treatment.
End-user Insights
-
Hospitals and Clinics dominate due to widespread usage in critical care and resuscitation.
-
Pharmaceutical Companies are rapidly adopting albumin for drug development and biologics, spurred by recombinant technologies and ethical production alternatives.
Regional Outlook
North America Leads with Strong Biotech Infrastructure
North America accounted for the largest market share in 2024, supported by advanced R&D capabilities, significant healthcare spending, and strong regulatory frameworks. NIH and CIHR funding programs continue to drive innovation in recombinant technologies and protein-based therapies.
Asia Pacific Shows Rapid Growth
The region is expected to witness the fastest growth, driven by:
-
Expanding biotech and pharmaceutical sectors in China, India, and South Korea.
-
A high prevalence of liver-related conditions, especially chronic hepatitis B in China.
-
Rising healthcare investments and growing demand for advanced therapies.
Recent Developments
-
May 2023: American Regent launched albumin-bound injectable suspension (alternative to Abraxane).
-
August 2022: Sartorius AG acquired Albumedix Ltd., boosting its recombinant albumin product offerings.
-
February 2022: Aadi Bioscience released Fyarro, an albumin-bound sirolimus-based cancer therapy.
Key Market Players
-
Octapharma AG
-
Albumedix Ltd.
-
Biotest AG
-
Grifols SA
-
HiMedia Laboratories
-
Medxbio Pte Ltd.
-
CSL Limited
-
Merck KGaA
-
Takeda Pharmaceutical Company Limited
-
Ventria Bioscience Inc.
Segmentation Overview
By Product
-
Human Albumin
-
Bovine Albumin
-
Recombinant Albumin
By Application
-
Drug Formulation
-
Therapeutics
-
Others
By End-user
-
Hospitals and Clinics
-
Pharmaceutical Companies
-
Research Institutions
By Geography
-
North America
-
Europe
-
Asia Pacific
-
Latin America
-
Middle East & Africa
For media inquiries, discounts, or customization requests, please contact us at sales@precedenceresearch.com