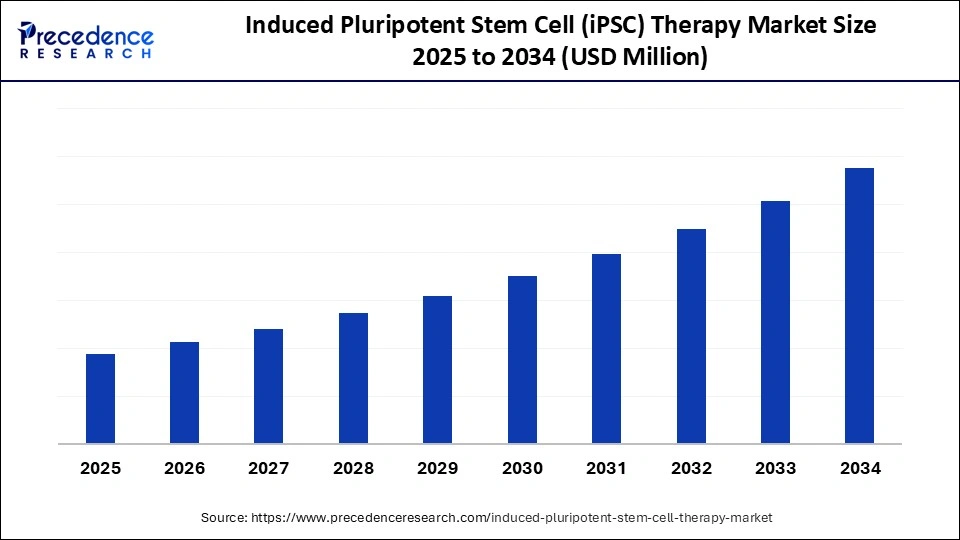
Global iPSC Therapy Market to Reach New Valuation by 2034, Fueled by a 12-15% CAGR and Pioneering Advances in Regenerative Medicine
The global induced pluripotent stem cell (iPSC) therapy market is positioned at the forefront of regenerative medicine and personalized healthcare, poised to witness robust growth at a projected CAGR of 12-15% between 2025 and 2034.
Key growth drivers include surging chronic disease prevalence, aging populations, and substantial R&D investment, alongside pivotal breakthroughs in reprogramming and scalable cell production platforms. The incorporation of artificial intelligence (AI) and machine learning is further propelling efficiency, clinical translation, and personalized therapeutic environments in this high-growth market.
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/7021
Induced Pluripotent Stem Cell (iPSC) Therapy Market Key Insights
-
The global iPSC therapy market is experiencing strong expansion, attributed to stem-cell research funding, expanding clinical pipelines, and patient-specific regenerative treatments.
-
North America is the dominating region, with over 40% market share in 2024 and a vibrant research and investment ecosystem; Asia Pacific is the fastest-growing segment, driven by advancing policy, funding, and clinical research.
-
Hematopoietic cells led by nearly 40% segment share in 2024, while iPSC-derived hepatocytes are expected to register the fastest CAGR due to exceptional demand for liver disease therapies.
-
Pharmaceutical and biotech firms represent around 50% of market revenue, relying on extensive R&D, strategic partnerships and rapid commercialization.
-
Leading market players include Fujifilm Cellular Dynamics, Astellas, Thermo Fisher Scientific, Takara Bio, Lonza, Merck, Bio-Techne, StemCell Technologies, and Cellular Dynamics International.
-
The regenerative medicine application segment contributed roughly 35% of total market value in 2024.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Revenue Breakdown Table
| Segment | Market Share (2024) | Fastest Growing Region | Notable Winners |
|---|---|---|---|
| Hematopoietic Cells | 40% | Asia Pacific | Fujifilm, Takara Bio |
| iPSC-Derived Cells | 45% | North America | Thermo Fisher, Astellas |
| Regenerative Medicine Application | 35% | Europe | Lonza, Merck |
| Pharma/Biotech End-Users | 50% | North America | Bio-Techne, StemCell |
| Stem Cell Banking Services | Fastest CAGR | Asia Pacific | Lonza, StemCell |
| Reprogramming Segment | 30% | North America | Thermo Fisher |
Artificial intelligence (AI) is radically transforming the iPSC therapy landscape by accelerating disease modeling, drug discovery, and patient outcome prediction. Advanced algorithms streamline high-throughput screening and optimize cell culture conditions for large-scale, efficient production. AI-driven data analysis also enables deeper insights into genetic and omics data, improving patient stratification and driving precision medicine forward.
Additionally, AI integration ensures enhanced reproducibility and quality control in iPSC workflows, allowing research teams to automate cell characterization and tackle complex multi-omics datasets. This push toward digitalization means faster discovery cycles, more robust safety profiles, and the ability to personalize cellular environments for disease modeling and pharmaceutical testing.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Growth Factors
-
Rising burden of chronic diseases worldwide, especially among aging populations, creates demand for innovative treatments and personalized medicine.
-
Technological breakthroughs in cell reprogramming and differentiation improve scalability and purity of iPSC-derived therapeutics.
-
Increasing public and private investment, particularly in North America, Asia Pacific, and Europe, catalyzes clinical translation and commercialization.
-
Strategic collaborations and alliances between biopharma leaders and specialty iPSC platform companies foster continuous innovation.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Opportunities and Trends
What Drives the Shift Toward Allogeneic “Off-the-Shelf” Cell Therapies?
Recent developments have spotlighted allogeneic therapies, utilizing donor-derived cells modified to minimize immune rejection, as viable solutions for mass production and accessibility. These universal therapies reduce logistical complexity and manufacturing timelines, making regenerative medicine more scalable, though patient-specific approaches still offer lower immunogenic risk.
How Are iPSCs Unlocking New Frontiers in Disease Modeling and Drug Discovery?
iPSCs provide patient-specific, human-relevant cellular models that hasten drug discovery and reduce reliance on animal testing. Their versatility in modeling complex conditions – from neurological to metabolic disorders – means higher translational accuracy and enables tailored therapeutic intervention.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Regional and Segment Analysis
North America: Market Powerhouse
North America led with 40% market share in 2024, driven by extensive NIH funding, a strong regulatory environment, and concentration of major biopharma players. The U.S. dominates with advanced infrastructure and robust federal support, making it the nucleus of global iPSC commercialization.
Asia Pacific: Fastest-Growing Region
Asia Pacific, particularly Japan, China, and India, is the fastest-growing region due to strong government funding, favorable regulations, and intense R&D in cell reprogramming and clinical trials. Japan’s PMDA accelerates approvals, while Chinese and Indian investments address high local demand for regenerative therapy.
Europe: Rigorous Regulatory Leadership
Europe’s EMA ensures standardized methods and collaborative innovation through public-private partnerships like EBiSC, with a noteworthy market value of $546 million in 2024.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Major Segments
-
By Cell Type: Hematopoietic cells (40% share), hepatocytes (fastest growth).
-
By Application: Regenerative medicine (35% share), disease modeling (fastest growth).
-
By Product & Services: Stem cell banking, GMP manufacturing, and cell characterization.
-
By Workflow: Reprogramming (30% share), cell characterization (fastest CAGR).
-
By End-User: Pharmaceutical/biotech (50% share), CRO/CDMOs (fastest CAGR).
Notable Company Breakthroughs & Major Market Players
-
Fujifilm Cellular Dynamics: Pioneer in iPSC-derived cells for drug discovery and regenerative therapy.
-
Astellas: Advancing blindness and neurodegenerative disease therapies with iPSC platforms.
-
Thermo Fisher Scientific: Leading in cell production, research tools, and clinical transitions.
-
Takara Bio: Innovative reprogramming and differentiation solutions powering clinical applications.
-
Lonza: Scalable iPSC platforms and GMP manufacturing for global reach.
-
Merck, Bio-Techne, Stemcell Technologies, Cellular Dynamics International: Supplying reagents, assay kits, and advanced research tools for global adoption.
Induced Pluripotent Stem Cell (iPSC) Therapy Market Challenges and Cost Pressures
The iPSC market faces critical challenges, notably the risk of tumorigenicity due to reprogramming processes and the need for stringent quality control. Cost pressures also arise from the complexities of GMP-compliant manufacturing, regulatory compliance, and the high overheads of personalized therapy development. Allogeneic (universal donor) approaches, while promising, remain expensive to scale and lack proven long-term immune compatibility.
Case Study Example
In the U.S., NIH-funded research led to the clinical translation of iPSC treatments for macular degeneration, resulting in improved visual outcomes and establishing a path for FDA-regulated trials in Parkinson’s and heart failure, demonstrating the pipeline’s potential.
Read Also: Pharmaceutical Blister Packaging Market
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344