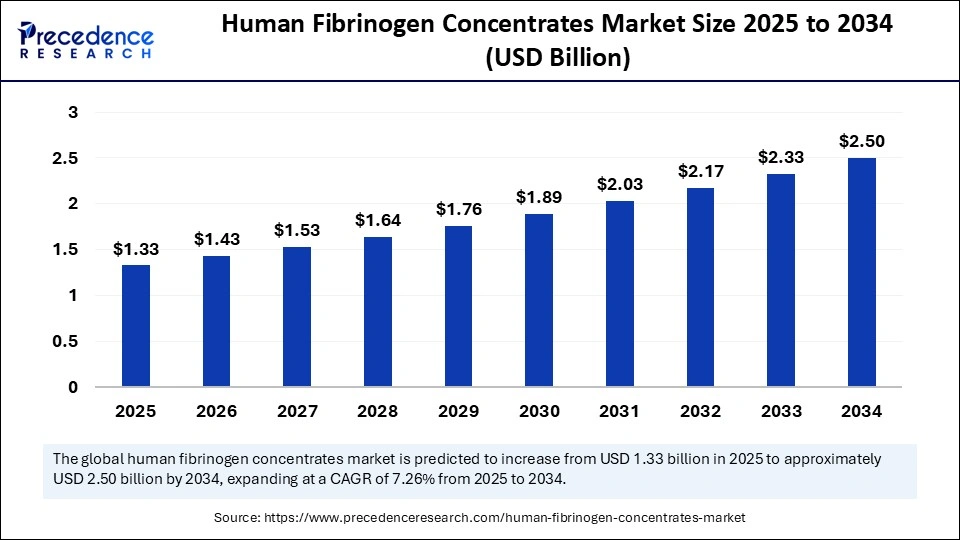
The global human fibrinogen concentrates market is expanding steadily, with a projected valuation of USD 1.33 billion in 2025 and an expected growth to approximately USD 2.50 billion by 2034. This growth is fueled by rising surgical procedures, increasing prevalence of bleeding disorders, and advancements in biotechnology, underpinning a compound annual growth rate (CAGR) of 7.26% from 2025 to 2034.
What is Driving the Growth of the Human Fibrinogen Concentrates Market?
The global market for human fibrinogen concentrates is witnessing robust growth, primarily driven by the increased number of surgeries and bleeding disorder cases worldwide. These concentrates, including plasma-derived and recombinant types, are essential in managing congenital and acquired fibrinogen deficiencies, as well as surgical and trauma-related bleeding. Enhanced awareness of rare coagulation disorders coupled with advances in biotechnology have further accelerated adoption, positioning fibrinogen concentrates as a critical therapeutic tool in contemporary medicine.
Human Fibrinogen Concentrates Market Key Insights
- The market size is projected at USD 1.33 billion in 2025, reaching USD 2.50 billion by 2034.
-
North America led the market with a 48% share in 2024.
-
Asia Pacific is the fastest-growing region, expected to expand at a CAGR of 9.5% through 2034.
-
Plasma-derived fibrinogen concentrates held the largest segment share of 64.4% in 2024.
-
Recombinant fibrinogen concentrates are the fastest growing segment, with a 10.4% CAGR.
-
Congenital fibrinogen deficiency represents the largest application segment with 42.4% market share.
-
Hospital pharmacies dominate distribution with 52.5% market share, while online pharmacies grow at 10%.
-
Hospitals are the largest end-users, accounting for 47.4% of demand in 2024.
Human Fibrinogen Concentrates Market Revenue and Forecast Tables
| Year | Market Size (USD Billion) | CAGR (%) |
|---|---|---|
| 2025 | 1.33 | 7.26 |
| 2026 | 1.43 | |
| 2034 | 2.50 |
Moreover, AI-driven data analytics support clinical decision-making by predicting patient-specific fibrinogen needs, personalizing dosage regimens, and improving treatment outcomes. These technological integrations accelerate time-to-market for novel fibrinogen therapies and facilitate continuous innovation in delivery systems, such as ready-to-use liquid concentrates, vital for emergency and surgical care scenarios.
What Factors Are Propelling Market Growth?
The rising volume of complex surgical interventions and prevalence of bleeding disorders globally are key drivers. Growing awareness and diagnosis of congenital fibrinogen deficiencies have increased the adoption of advanced fibrinogen therapies in clinical practice.
Technological strides in plasma fractionation and recombinant fibrinogen production are expanding supply, improving safety profiles, and enabling rapid administration in acute care. Additionally, the shift from traditional cryoprecipitate to fibrinogen concentrates for hemostasis management is significantly broadening market opportunities.
What Opportunities and Trends Are Emerging in This Market?
Is recombinant technology reshaping the future of fibrinogen therapies?
Recombinant fibrinogen concentrates are gaining traction due to their pathogen-free production process and scalability, reducing dependency on plasma donors and enhancing safety. This segment is projected to grow at a CAGR of 10.4%, driven by biotechnology innovations and regulatory support.
How are emerging economies influencing market dynamics?
Emerging markets in Asia Pacific, notably China and India, are witnessing surging demand due to rising healthcare investments, increasing surgical procedures, and regulatory facilitation. These regions offer significant growth potential owing to expanding healthcare infrastructure and large patient populations.
Are digital channels disrupting fibrinogen concentrate distribution?
Online pharmacies are emerging as critical distribution channels, growing at a 10% CAGR. The integration of digital health platforms is streamlining supply chains and improving patient access, particularly in critical care therapies.
Regional and Segmentation Analysis
North America dominates due to aging populations, robust healthcare infrastructure, and high prevalence of coagulation disorders.
Asia Pacific’s rapid growth is boosted by increasing surgical volume and government health initiatives.
Europe’s market benefits from advanced plasma fractionation facilities and strategic investments from major industry players.
Segment-wise, plasma-derived fibrinogen concentrates lead the market, while recombinant types are fastest growing.
Congenital fibrinogen deficiency remains the primary application area, and hospital pharmacies alongside hospitals hold dominant shares in distribution and end-use respectively.
Challenges and Cost Pressures
Despite growth prospects, the market faces challenges such as high production costs, regulatory complexities, and the need for cold chain logistics. Ensuring consistent supply of plasma-derived materials and scaling recombinant technologies cost-effectively remain critical hurdles. Market players must navigate evolving reimbursement landscapes and competition from alternative hemostatic agents.
Top Companies Operating in the Human Fibrinogen Concentrates Market
Tier I — Major Players
These are the dominant, globally scaling companies that lead innovation, distribution, and regulatory presence. Together, they account for around 40 50% of global revenue in this niche.
- CSL Behring: It is a global leader in plasma-derived therapies, offering FDA-approved fibrinogen concentrates like RiaSTAP®, and driving innovation through continuous investment in R&D and global plasma collection infrastructure.
- Octapharma AG: The company plays a significant role through its product Fibryga®, which is widely used for both congenital and acquired fibrinogen deficiencies, supported by strong clinical trials and global distribution channels.
- LFB Group: It contributes with its fibrinogen product FibCLOT®, focused on treating rare bleeding disorders, particularly in Europe, and is recognized for its emphasis on quality plasma fractionation and regulatory compliance.
Tier II — Mid Level Contributors
These firms have strong market presence, often regionally or in specialized product lines, but aren’t as dominant as Tier I. Collectively, they contribute ~30 35%.
- Grifols, S.A.: It leverages its global plasma collection network and advanced fractionation technologies to produce high-purity fibrinogen concentrates, supporting both clinical and surgical applications, especially in Europe and North America.
- Shanghai RAAS Blood Products Co., Ltd.: It is a key player in China’s plasma-derived product market, supplying fibrinogen concentrates to meet growing domestic demand, backed by government support and expanding healthcare infrastructure.
- Baxter International: The company contributes through its historical expertise in blood and plasma therapies, and while not a leading fibrinogen brand owner, it maintains a strong presence in the broader hemostatic and surgical bleeding management market.
Tier III — Niche & Regional Players
These are smaller, localized, or specialized companies whose contributions are modest individually, but cumulatively make up ~15 20%.
- Hualan Biological Engineering Inc.: The company plays a significant role in China’s plasma-derived therapeutics sector, producing human fibrinogen concentrates for domestic clinical use, with a focus on expanding R&D and GMP-compliant manufacturing capacity.
- Jiangxi Boya Bio Pharmaceutical: It contributes to the Chinese market with its portfolio of plasma-derived products, including fibrinogen concentrates, catering to rising regional demand for bleeding disorder treatments and surgical hemostasis.
- Green Cross Corporation (now GC Pharma): It is a major South Korean biopharmaceutical firm, supplies fibrinogen concentrates domestically and across select global markets, leveraging its advanced plasma fractionation and regulatory compliance capabilities.
- Harbin Pacific Biopharmaceutical Co., Ltd.: It focuses on plasma product innovation and manufacturing within China, contributing to the market by producing fibrinogen concentrates that support clinical and surgical use under national health initiatives.
Case Study: Octapharma’s National Clinical Trial Initiative
In July 2025, Octapharma secured $29 million in funding for a national clinical trial led by the University of Colorado Anschutz Medical Campus. The study evaluates early fibrinogen replacement therapy in trauma patients with life-threatening bleeding. This initiative underscores the clinical importance and investment potential in advancing fibrinogen concentrate therapies for critical care.
Read Also: Flash Chromatography Market
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344